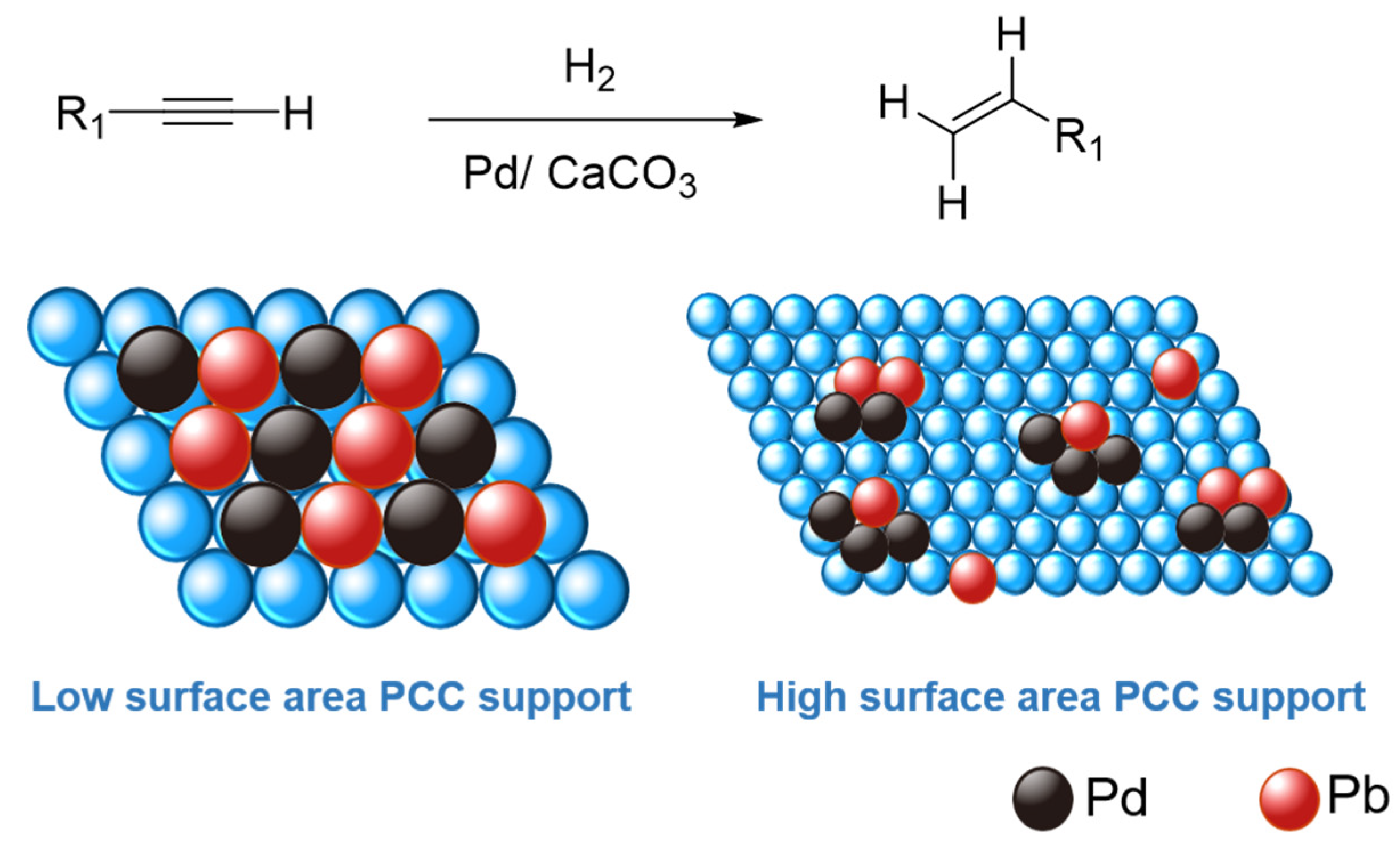
Catalysts | Free Full-Text | Revisiting the Semi-Hydrogenation of Phenylacetylene to Styrene over Palladium-Lead Alloyed Catalysts on Precipitated Calcium Carbonate Supports
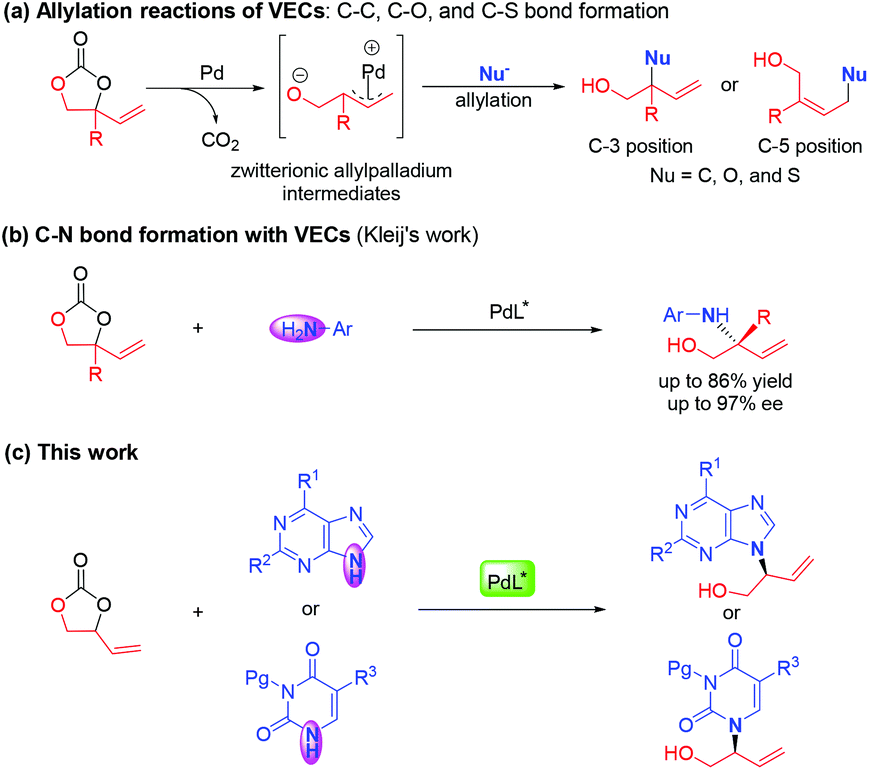
Palladium-catalyzed asymmetric allylic amination of a vinylethylene carbonate with N-heteroaromatics - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00272D

An efficient palladium-catalyzed synthesis of benzils from aryl bromides: vinylene carbonate as a synthetic equivalent of glyoxal - ScienceDirect

Palladium-catalyzed oxyarylation of olefins using silver carbonate as the base. Probing the mechanism by electrospray ionization mass spectrometry - ScienceDirect
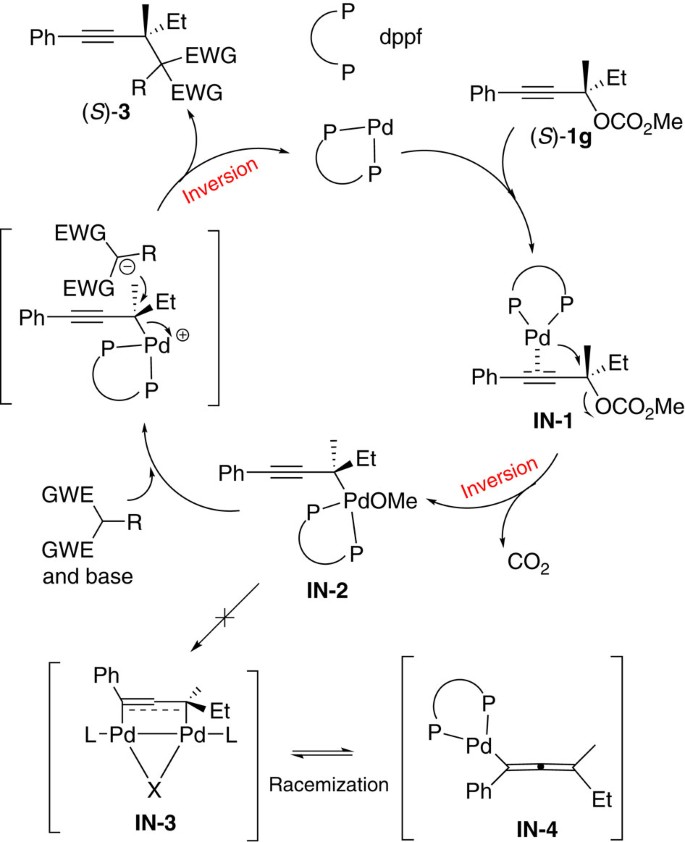
Palladium-catalysed formation of vicinal all-carbon quaternary centres via propargylation | Nature Communications