
ArtStation - Plasma Battery & Battery Container - Concept, Debora Spiro | Plasma, Tech design, Concept
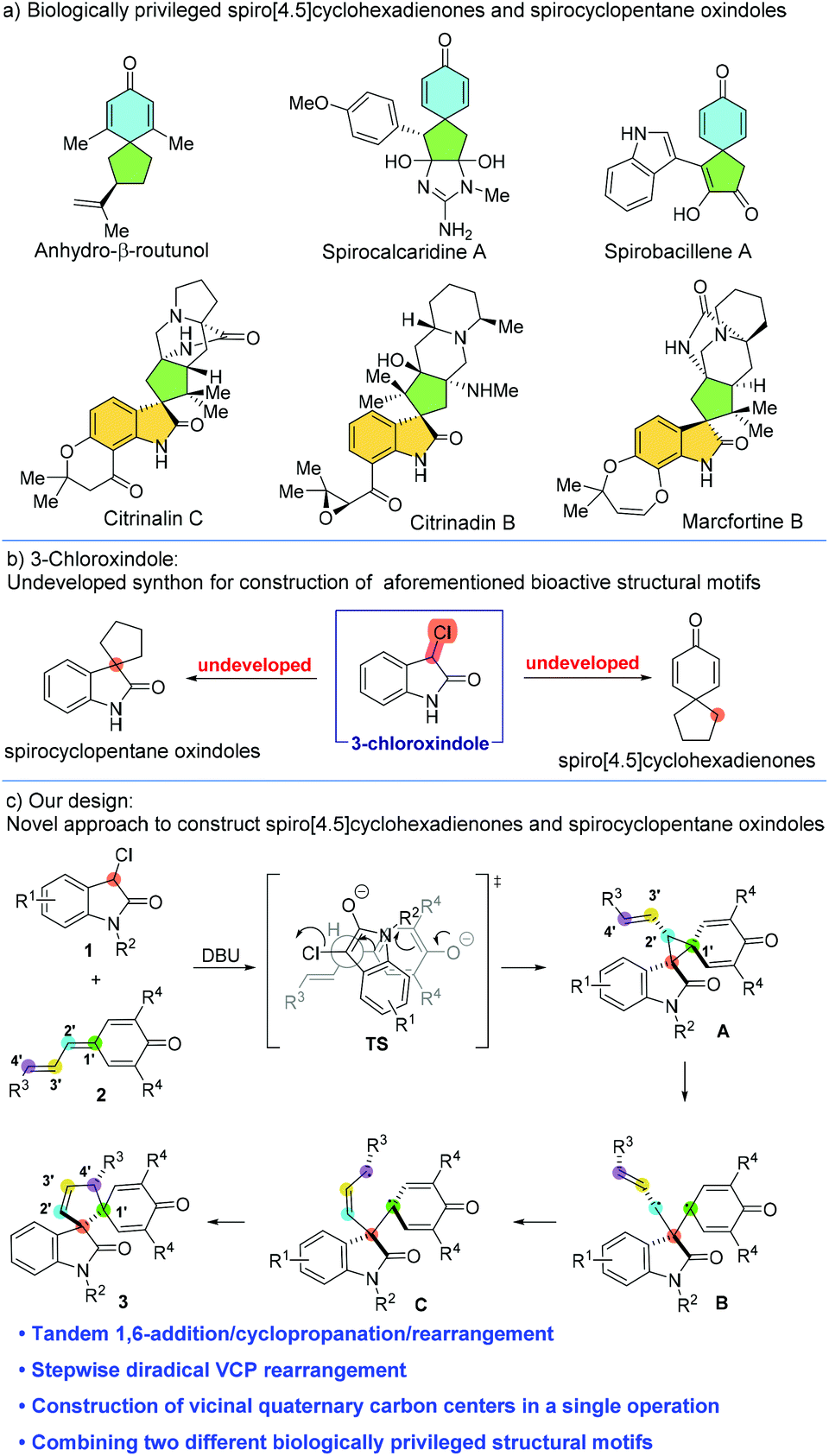
Tandem 1,6-addition/cyclopropanation/rearrangement reaction of vinylogous para -quinone methides with 3-chlorooxindoles: construction of vicinal quate ... - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D2QO00471B
![Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.8b00518/asset/images/medium/ol-2018-00518f_0003.gif)
Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters
![Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.8b00518/asset/images/medium/ol-2018-00518f_0009.gif)
Cascade Radical 1,6-Addition/Cyclization of para-Quinone Methides: Leading to Spiro[4.5]deca-6,9-dien-8-ones | Organic Letters

Crystals | Free Full-Text | Penetration Gain Study of a Tungsten-Fiber/Zr-Based Metallic Glass Matrix Composite
![Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B](https://pubs.rsc.org/image/article/2015/OB/c5ob01077b/c5ob01077b-s1_hi-res.gif)
Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B
![Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B](https://pubs.rsc.org/image/article/2015/OB/c5ob01077b/c5ob01077b-f1_hi-res.gif)
Domino Prins/pinacol reaction for the stereoselective synthesis of spiro[pyran-4,4′-quinoline]-2′,3′-dione derivatives - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01077B

Oxidative Biaryl Coupling Reaction of Phenol Ether Derivatives Using a Hypervalent Iodine(III) Reagent | The Journal of Organic Chemistry
![Silver-Catalyzed Cascade 1,6-Addition/Cyclization of para-Quinone Methides with Propargyl Malonates: An Approach to Spiro[4.5]deca-6,9-dien-8-ones | The Journal of Organic Chemistry Silver-Catalyzed Cascade 1,6-Addition/Cyclization of para-Quinone Methides with Propargyl Malonates: An Approach to Spiro[4.5]deca-6,9-dien-8-ones | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.7b01400/asset/images/medium/jo-2017-01400j_0002.gif)
Silver-Catalyzed Cascade 1,6-Addition/Cyclization of para-Quinone Methides with Propargyl Malonates: An Approach to Spiro[4.5]deca-6,9-dien-8-ones | The Journal of Organic Chemistry











