Design and synthesis of simplified speciophylline analogues and β‐carbolines as active molecules against Plasmodium falciparum - Pierrot - 2019 - Drug Development Research - Wiley Online Library

Metal‐Free Synthesis of N‐Aryl Amides using Organocatalytic Ring‐Opening Aminolysis of Lactones - Guo - 2017 - ChemSusChem - Wiley Online Library

Triazabicyclodecene: An Effective Isotope Exchange Catalyst in CDCl3 | The Journal of Organic Chemistry
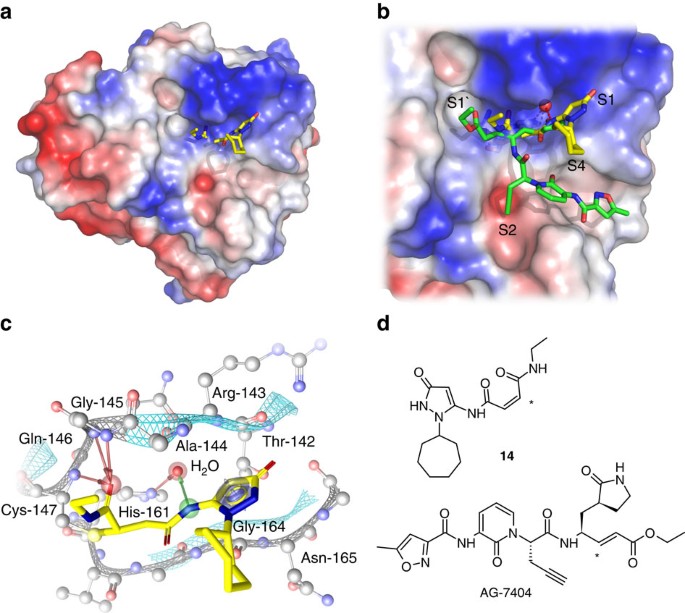
Irreversible inhibitors of the 3C protease of Coxsackie virus through templated assembly of protein-binding fragments | Nature Communications

Identification of isoform/domain-selective fragments from the selection of DNA-encoded dynamic library - ScienceDirect
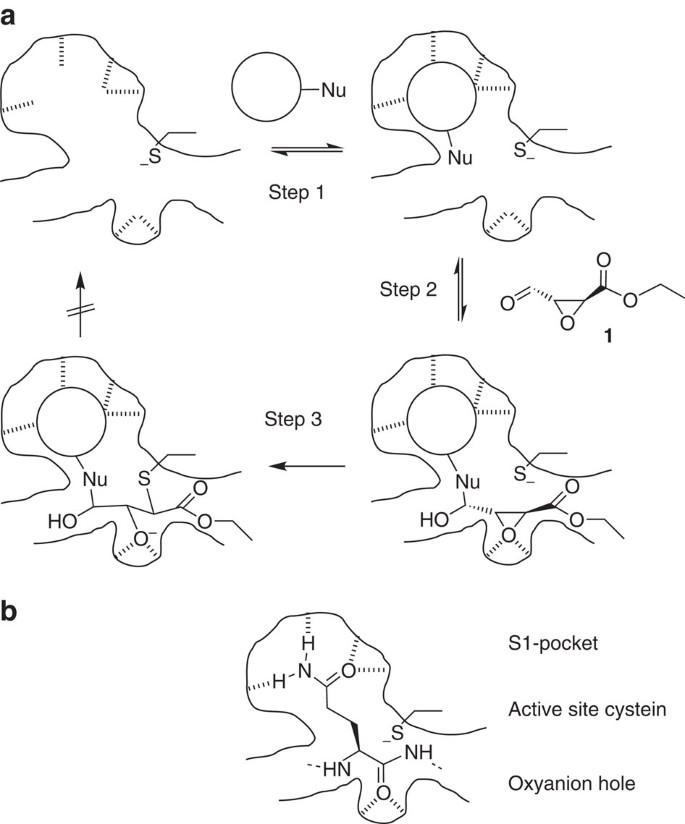
Irreversible inhibitors of the 3C protease of Coxsackie virus through templated assembly of protein-binding fragments | Nature Communications

US8834445B2 - Methods of treating or preventing peritonitis with oxidative reductive potential water solution - Google Patents

Traceless and Chemoselective Amine Bioconjugation via Phthalimidine Formation in Native Protein Modification | Organic Letters

Synthesis of 3‐Oxaterpenoids and Its Application in the Total Synthesis of (±)‐Moluccanic Acid Methyl Ester - Li - 2012 - Angewandte Chemie - Wiley Online Library
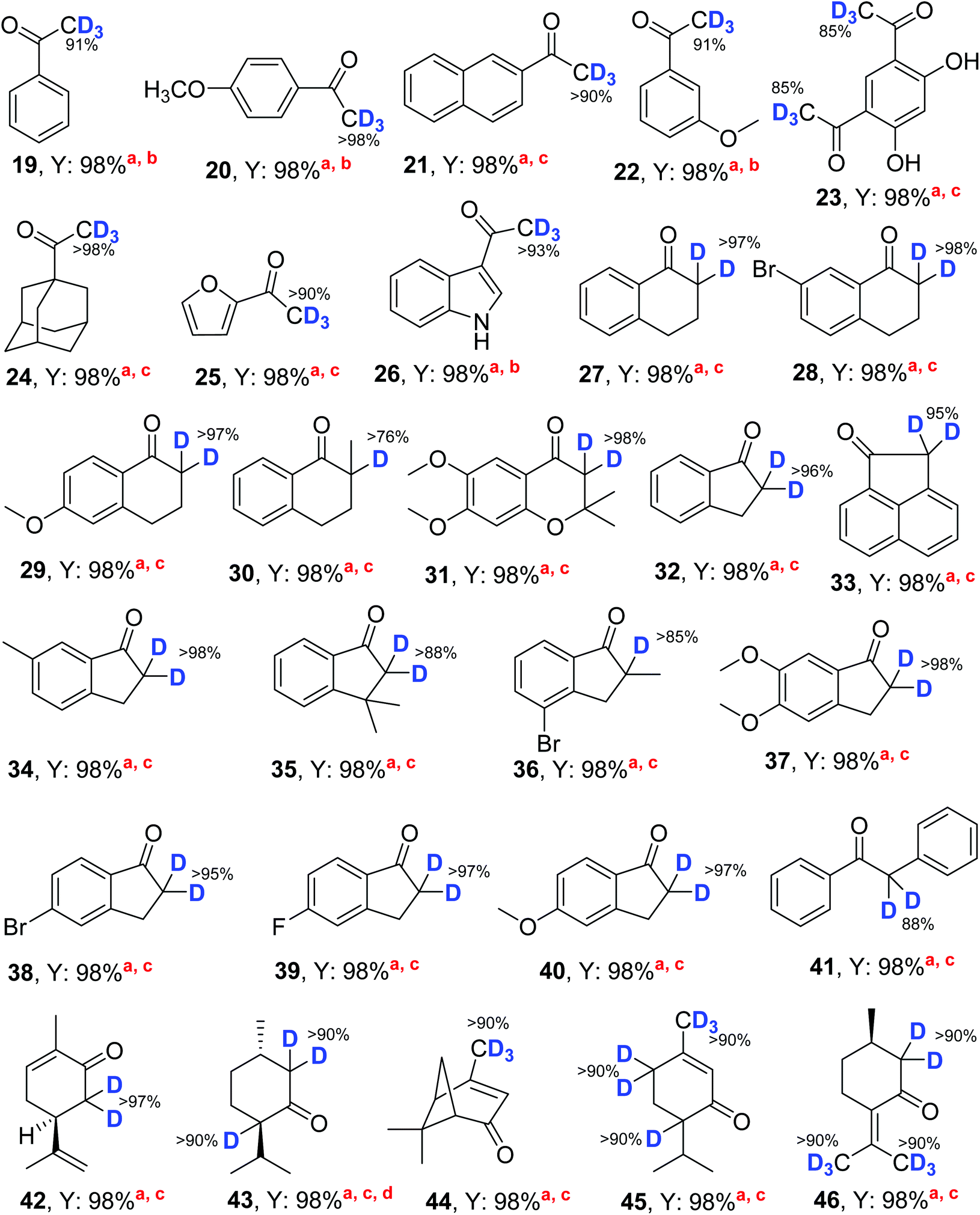
Spontaneous conversion of prenyl halides to acids: application in metal-free preparation of deuterated compounds under mild conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB01275D

Materials | Free Full-Text | Controlling Shock-Induced Energy Release Characteristics of PTFE/Al by Adding Oxides | HTML

Protein‐Templated Fragment Ligations—From Molecular Recognition to Drug Discovery - Jaegle - 2017 - Angewandte Chemie International Edition - Wiley Online Library