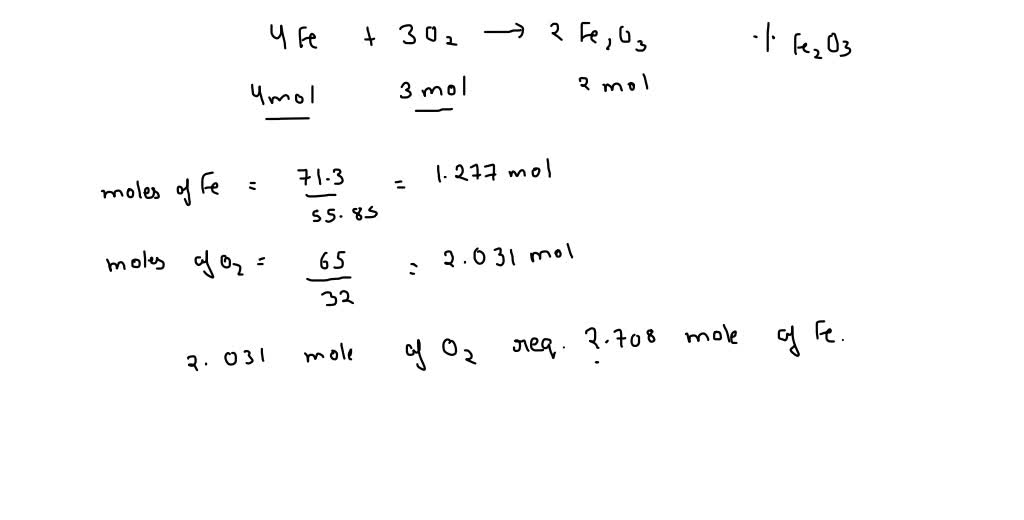
SOLVED: 4Fe(s)+3O2(g)⟶2Fe2O3(s) If 71.3 g of Fe and 65.0 g of O2 are mixed, and 85.0 g of Fe2O3 is actually obtained, what is the percent yield of Fe2O3 for the reaction?

XRF pattern of ocher particles: 1-FeO(OH) goethite; 2-Fe2O3 hematite;... | Download Scientific Diagram
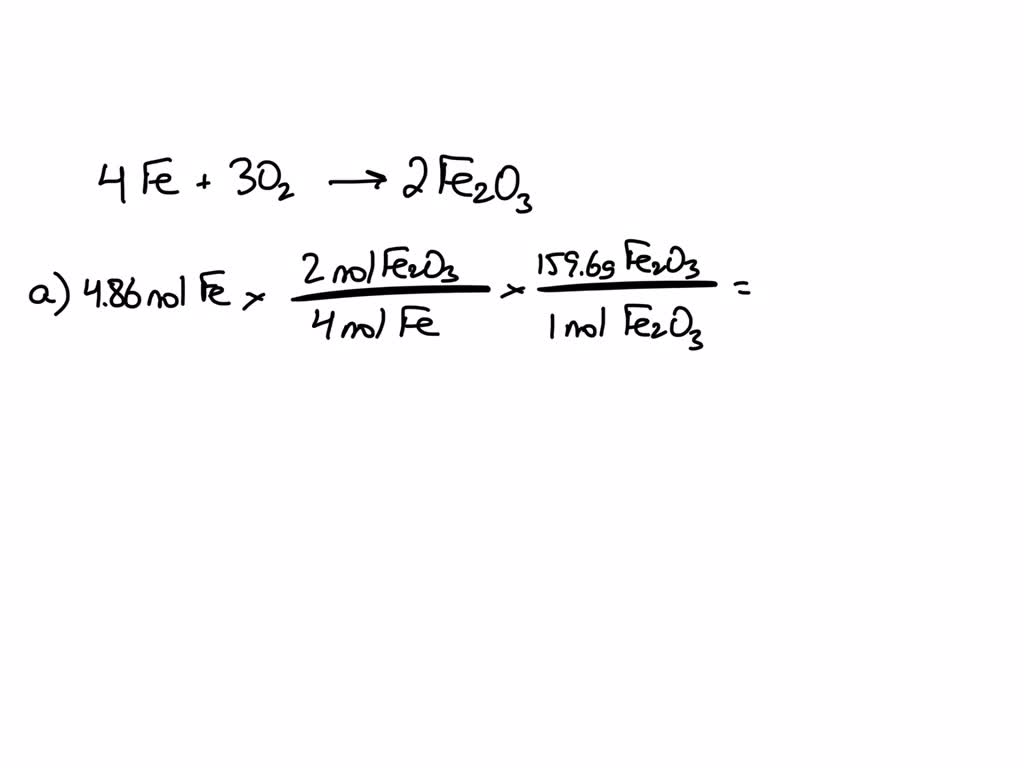
SOLVED: Given the reaction 4Fe + 3O2= 2Fe2O3 a. How many grams of Fe2O3 will be formed from 4.86 moles Fe reacting with sufficient oxygen gas? b. How many grams of Fe












![Perspective view of two isomers of [Fe4(mdea)6]•6CHCl3: (a) the... | Download Scientific Diagram Perspective view of two isomers of [Fe4(mdea)6]•6CHCl3: (a) the... | Download Scientific Diagram](https://www.researchgate.net/publication/287799370/figure/fig9/AS:669689801875466@1536677899349/Perspective-view-of-two-isomers-of-Fe4mdea66CHCl3-a-the-chair-shaped-isomer-1a.png)

